 
Learn how Bohr's model calculated the energy levels, radii, and emission lines of hydrogen and other atoms. It explains the discrete line spectrum of hydrogen emission by quantizing the energy levels of electrons and photons. + - n 3 n 2 n 1 Prepared by: Sidra Javed. 2- As long as an electron remain in a shell it never gains or losses energy. In doing so, it emits a photon of energy equal to the difference in its energies in the two orbits. Bohr's model of hydrogen is based on the nonclassical assumption that electrons travel in specific shells, or orbits, around the nucleus. Bohr’s Model of Atom 1- Electrons revolves around the nucleus in definite energy levels called orbits or shells in an atom without radiating energy. An electron can make a transition from one of its orbits to another orbit having lower energy.Today, we know that the Bohr Model has some inaccuracies, but it’s still used because of its simple approach to. Bohrs model kunne både forklare hydrogens spektrum, Rutherfords påvisning af atomkernen og andre problemer med modstrid mellem den klassiske fysik og nyere eksperimenter. 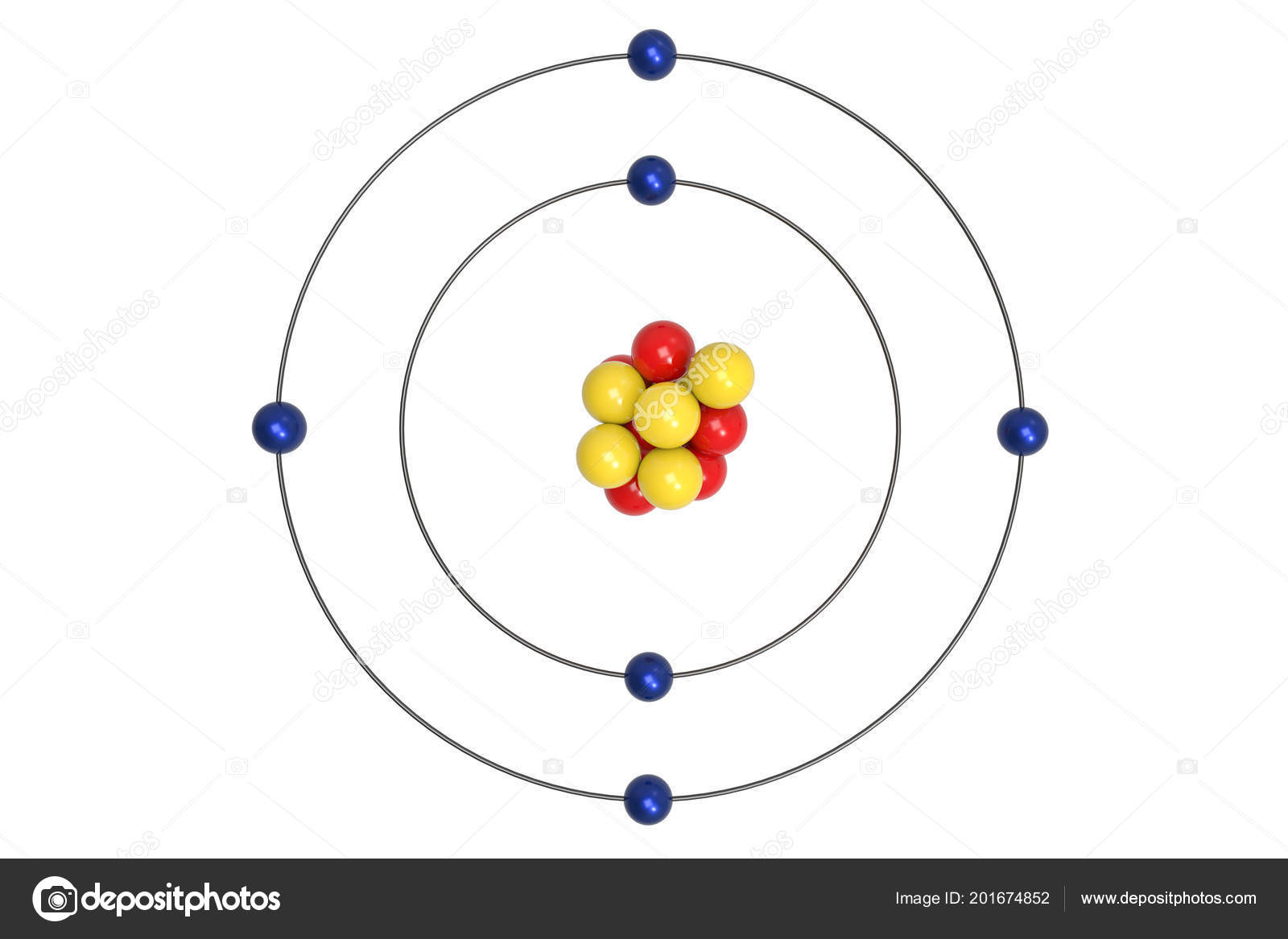
The Bohr Model has an atom with a positively-charged nucleus surrounded by negatively-charged electrons that have circular, planetary-like orbits. Thus, different orbits have different and definite values of angular momentum and therefore, different values of energy. The Bohr Model is a modification of an earlier atomic model, the Rutherford Model. Such orbits are called stable orbits or stable states of the electrons and electrons in these orbits do not emit radiation as is demanded by classical physics. The radius of the orbit of an electron can only take certain fixed values such that the angular momentum of the electron in these orbits is an integral multiple of `"h"/(2π)`, h being Planck’s constant.This is the same assumption as in Rutherford’s model and the centripetal force necessary for the circular motion is provided by the electrostatic force of attraction between the electron and the nucleus. The electrons revolve around the nucleus in circular orbits.The postulates of Bohr's atomic model (for the hydrogen atom): The postulates of Bohrs atomic model (for the hydrogen atom): The electrons revolve around the nucleus in circular orbits. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
